Photobiological Eyewear India: Sleepaxa's Complete Clinical Guide (2026) | Migraine & Sleep Glasses
Photobiological Eyewear India: Sleepaxa's Complete Clinical Guide
How wavelength-selective filtration targets migraine photophobia at the ipRGC level and protects circadian melatonin rhythms — India's first comprehensive clinical resource on photobiological eyewear.
Introduction: Why India Urgently Needs Photobiological Eyewear
India is in the grip of a silent neurological epidemic. Population-based studies from Karnataka (Kulkarni et al., J Headache Pain) and Delhi NCR (Chowdhury et al., 2024) consistently report a one-year migraine prevalence of 25–26% among Indian adults — nearly double the global average of 14.7%. A 2025 PMC scoping review estimated approximately 213 million active migraine cases annually in India, with productivity losses of 17.3 days per person per year, translating to an economic burden of approximately ₹18,674 crore (USD 22.21 billion) annually.
Yet until 2024, India had zero domestic photobiological eyewear companies. Individuals seeking wavelength-selective migraine glasses or scientifically calibrated sleep glasses had to rely on expensive imports from international brands like Avulux (USA), TheraSpecs (USA), or Axon Optics — costing ₹8,000–15,000+ with no local clinical support, no Indian patent protection, and no customisation for the Indian consumer.
Sleepaxa Private Limited was founded to close this gap. As India's first photobiological eyewear company, Sleepaxa combines deep optical engineering, polymer chemistry, circadian neuroscience, and clinical optometry expertise to develop eyewear that doesn't just "block blue light" — it selectively manages the precise wavelength bands that peer-reviewed neuroscience has identified as the triggers for migraine-associated photophobia and circadian melatonin suppression. Every product is developed under the clinical oversight of Sleepaxa's Clinical Advisory Board and made accessible for the Indian market.
In this comprehensive clinical guide, we cover the neuroscience of photobiology, explain Sleepaxa's patented NeuroCalm FLX+™ technology and patent-pending Circadian560™ technology, detail our complete product range, and provide the full clinical evidence base. Whether you're a healthcare professional, a migraine sufferer, or someone seeking better sleep — this is the most complete resource on photobiological eyewear in India.
1. What Is Photobiological Eyewear? A Complete Explanation
1.1 Core Concept: Wavelength-Selective Filtration vs. Simple Light Reduction
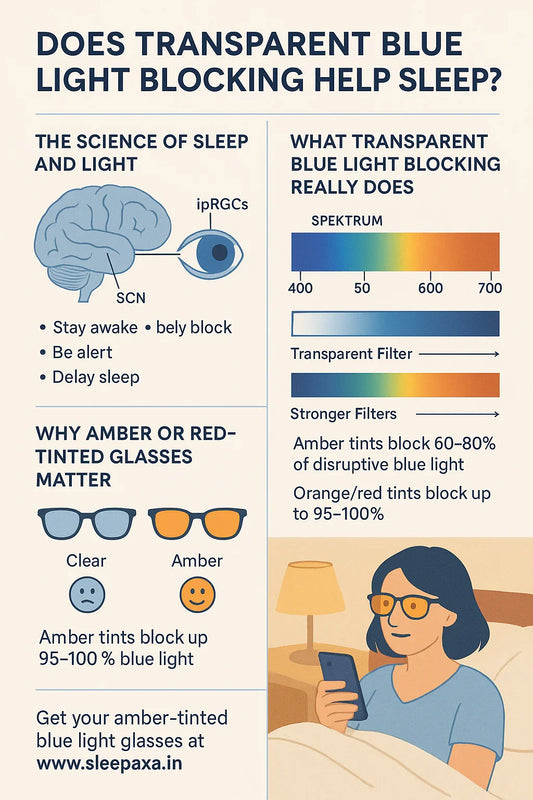
Photobiological eyewear is built on the science of photobiology — the interdisciplinary study of how electromagnetic radiation in the visible spectrum (380–750 nm) interacts with biological systems including the retina, neuroendocrine pathways, and the central nervous system. The key principle is wavelength selectivity: rather than uniformly reducing all light (as sunglasses do), photobiological lenses attenuate specific nanometre bands that scientific evidence has identified as biologically problematic, while allowing beneficial wavelengths to pass through.
This distinction matters enormously. Wearing overly dark lenses indoors causes dark adaptation — a neuroplastic process where the visual system recalibrates to lower light levels, paradoxically increasing photosensitivity over time. The American Academy of Ophthalmology and multiple neuro-ophthalmologists have cautioned against habitual indoor dark-lens use for this reason. Photobiological eyewear avoids this trap by maintaining adequate overall visible light transmission while selectively filtering only the wavelengths that trigger pathological responses.
1.2 The Neuroscience: ipRGCs, Melanopsin, and the Trigeminal Pathway
The ipRGC Discovery That Changed Everything
In 2002, Hattar et al. (Science) and Berson et al. identified a specialised class of retinal neurons — intrinsically photosensitive retinal ganglion cells (ipRGCs) — that contain the photopigment melanopsin. Unlike classical rods and cones responsible for image-forming vision, ipRGCs serve non-image-forming (NIF) functions. Melanopsin has peak spectral sensitivity at approximately 480 nm (blue-cyan light), though its effective activation range extends meaningfully from approximately 420–560 nm when accounting for in-vivo spectral broadening and chromatic adaptation.
These ipRGCs project directly to two critical brain targets. First, the suprachiasmatic nucleus (SCN) — the master circadian pacemaker that regulates melatonin secretion, cortisol rhythm, core body temperature, and the sleep-wake cycle. Second, the posterior thalamus, where ipRGC axons innervate neurons that also receive nociceptive (pain) input from dural trigeminal afferents (Noseda et al., Nature Neuroscience, 2010). This anatomical convergence is the neural mechanism explaining why light stimulation through ipRGCs can directly generate somatic pain — particularly in individuals with migraine.
Migraine Photophobia: Postretinal Amplification of ipRGC Signals
A landmark study from McAdams, Kaiser et al. published in Proceedings of the National Academy of Sciences (PNAS, 2020) fundamentally reshaped our understanding of migraine photophobia. By selectively stimulating melanopsin and cone photoreceptors independently, they demonstrated that people with migraine do not differ from healthy controls in how their retinas combine cone and melanopsin signals. Instead, migraine is associated with a postretinal amplification of the combined ipRGC signal specifically for the perception of discomfort — meaning the brain processes normal-intensity light as painfully uncomfortable.
Zele et al. (Cephalalgia, 2021) confirmed that melanopsin hypersensitivity dominates interictal photophobia in migraine. Their data showed melanopsin contributions to photophobia were approximately 1.5× greater than cone luminance contributions, with migraineurs demonstrating photophobia thresholds approximately 0.55 log units (3.5×) lower than controls. Nagata et al. (IJMS, 2024) extended this further, demonstrating that ipRGC hypersensitivity can induce cortical spreading depression (CSD) — the electrophysiological wave underlying migraine aura — and that CSD was suppressed by melanopsin inhibition.
Circadian Disruption: How Evening Light Destroys Sleep Architecture
The same ipRGC–melanopsin pathway driving migraine photophobia also regulates the circadian system. When blue light reaches ipRGCs in the evening, they signal the SCN to suppress pineal melatonin secretion — the hormone essential for sleep onset and circadian phase regulation. The Journal of Applied Physiology (West/Brainard et al., 2011) published the definitive dose-response curve: blue LED light (peak 469 nm) produces a sigmoidal, irradiance-dependent suppression of plasma melatonin with an ED50 of approximately 14.19 μW/cm². Harvard researchers confirmed blue light suppresses melatonin approximately twice as long as green light of comparable brightness and shifts circadian phase by 3 hours versus 1.5 hours.
Crucially, the systematic review by Tähkämö et al. (Chronobiology International, 2019) demonstrated that circadian sensitivity extends beyond the narrow blue band. Even green wavelengths (500–540 nm) and intermittent light exposures produce measurable circadian resetting responses. A 2025 study published in Life (MDPI) comparing red versus blue LED exposure over three hours found blue light maintained melatonin suppression at 7.5 pg/mL at the two-hour mark, while red light permitted recovery to 26.0 pg/mL (p = 0.019).
The 2025 Frontiers in Neurology meta-analysis of randomised controlled crossover trials on blue-light-blocking glasses confirmed that evening exposure to short-wavelength, blue-enriched light from electronic screens suppresses melatonin secretion, delays sleep onset latency, and alters circadian rhythms — providing the evidence base for wavelength-specific evening eyewear as a non-pharmacological chronobiological intervention.
Why this matters: Photobiological eyewear exists to selectively intervene at the wavelength level — reducing the specific light signals that ipRGCs use to trigger migraine-associated pain amplification and suppress evening melatonin. This is not about "blocking blue light" — it is about precision management of the melanopsin-activation spectrum.
2. Sleepaxa: India's Photobiological Eyewear Pioneer
2.1 R&D Leadership and Clinical Advisory Board
Every Sleepaxa lens technology is developed through a rigorous evidence-based pipeline — not trend-chasing or marketing-led formulation. Sleepaxa's R&D team brings together clinical optometry, spectrophotometric analysis, polymer-substrate engineering, and circadian neuroscience expertise.
Senior Optom. Suraj Dubey leads Sleepaxa's R&D as Head of Research and Development. His expertise spans clinical optometry, photobiological lens design, spectral curve optimisation, and optical polymer engineering. Suraj oversees the technical development of every Sleepaxa lens technology from concept through spectrophotometric validation to production-grade quality control.
Dr. Monica Choudhary serves as the Founding Member of Sleepaxa's Clinical Advisory Board. Dr. Choudhary provides clinical oversight across all product lines, ensuring each technology meets the clinical evidence standards expected by healthcare professionals. Her role bridges laboratory spectral engineering with real-world patient outcome expectations.
2.2 Brand Architecture: Sleepaxa as the Umbrella
Sleepaxa operates as the umbrella brand — analogous to how Apple houses distinct product technologies under one trusted name. Under the Sleepaxa umbrella sit proprietary sub-technologies, each addressing a distinct photobiological need. This ensures every product carries the full clinical authority and patent-backed credibility of the Sleepaxa name: Sleepaxa's NeuroCalm FLX+™ for migraine and photophobia, Sleepaxa's Circadian560™ for maximum sleep support, and a range of condition-specific eyewear built on the same scientific foundation.
3. Sleepaxa's Proprietary Technologies: Deep Dive
3.1 Sleepaxa's NeuroCalm FLX+™ — India's Only Patented Dual-Band Migraine Eyewear Technology
"An Optical Lens with Dual-Band Selective Light Attenuation"
Indian Patent Application No. IN 202521094270 · Grant Order Received · Innovation: Novel dual-band wavelength-selective optical filtering architecture for migraine and photophobia management
Why Dual-Band? The Limitation of Single-Band FL-41
Traditional FL-41 lenses — the current standard of care in photobiological migraine management — target a single wavelength band, primarily the 480–520 nm blue-green region where melanopsin sensitivity peaks. While effective (studies show 50%+ migraine frequency reduction in children and 76% photophobia improvement in adults), single-band FL-41 has a fundamental limitation identified by University of Utah researchers: melanopsin is bistable. This means the 11-cis retinal form of melanopsin (activated by ~480 nm light) is regenerated by longer-wavelength light, including orange-red wavelengths around 590–620 nm. The University of Utah's thin-film notch filter study (Hoggan et al., J Clinical Neuroscience, 2016) designed both a 480 nm therapeutic filter and a 620 nm filter intended as a sham — yet both produced clinically and statistically significant HIT-6 score reductions, precisely because the "sham" was blocking the melanopsin-regeneration wavelength.
Sleepaxa's NeuroCalm FLX+™ addresses this with a patented dual-band architecture that simultaneously attenuates two critical wavelength bands: Band 1 covers the primary melanopsin-activation zone (approximately 480–520 nm, where ipRGC sensitivity peaks and photophobia signalling originates); Band 2 targets the secondary wavelength range involved in melanopsin bistability and additional photophobic pathways, ensuring that the melanopsin chromophore cannot be efficiently regenerated after light-induced isomerisation. This dual-band approach reflects the current best understanding of photophobia neuroscience, providing a more complete intervention than any single-band lens can achieve.
Who Benefits from NeuroCalm FLX+™?
Chronic and episodic migraine sufferers experiencing interictal and ictal photophobia. Individuals with photophobia from post-concussion syndrome, traumatic brain injury (TBI), or benign essential blepharospasm. People experiencing visual discomfort under fluorescent or LED lighting at work, in hospitals, or in educational settings. Anyone seeking a non-pharmacological, evidence-based intervention for light-triggered discomfort as part of a comprehensive migraine management plan — complementing, not replacing, professional medical treatment. Explore Sleepaxa's NeuroCalm FLX+™ migraine glasses.
3.2 Sleepaxa's Circadian560™ — Patent-Pending Maximum Circadian Protection Technology
Maximum Blocking Up to 560 nm — Blue, Green & Partial Yellow Attenuation
Indian Patent Application No. IN 202521120977 · Purpose: Maximum circadian melatonin protection through comprehensive melanopic-range attenuation
Why 560 nm? Because "Blue" Is Only Half the Story
Most consumer "blue-light-blocking" glasses filter wavelengths up to 420–450 nm — missing the critical fact that melanopsin-driven melatonin suppression and circadian phase-shifting extend well into the green and short-yellow portions of the spectrum. The melanopsin spectral sensitivity function, when measured in vivo with pre-receptoral lens and macular pigment filtering, shows significant activation through at least 540 nm, with residual circadian influence detectable at even longer wavelengths. Harvard's research confirmed green light still shifted circadian phase by 1.5 hours — a clinically meaningful disruption that would not be prevented by standard blue-blocking glasses.
Sleepaxa's Circadian560™ takes this extended melanopic sensitivity to its logical conclusion: maximum blocking up to 560 nm. This covers the entire melanopic sensitivity range including blue (420–490 nm), green (490–540 nm), and partial yellow (540–560 nm). The result is what Sleepaxa's R&D team terms "Maximum Circadian Shield" — creating an optical environment that closely approximates the short-wavelength-depleted light conditions under which human circadian biology evolved (warm firelight after sunset), even while the user continues to work on screens or under artificial lighting.
Who Benefits from Circadian560™?
Anyone with sleep onset difficulty, delayed sleep phase syndrome, or irregular sleep patterns. Night-shift workers, healthcare professionals (doctors, nurses), and IT professionals needing circadian protection during evening work. Heavy evening screen users including students preparing for exams, gamers, and remote workers. Parents seeking a healthy, non-pharmacological screen-time wind-down tool for the family. Individuals with circadian-linked conditions where melatonin timing is clinically important. Explore Sleepaxa's Circadian560™ sleep glasses.
3.3 Technology Comparison: NeuroCalm FLX+™ vs. Circadian560™
| Parameter | NeuroCalm FLX+™ | Circadian560™ |
|---|---|---|
| IP Status | Patent Granted (IN 202521094270) | Patent Pending (IN 202521120977) |
| Mechanism | Dual-band selective attenuation | Extended-range blocking up to 560 nm |
| Primary Target | Migraine photophobia, trigeminal light sensitivity | Circadian melatonin rhythm, sleep architecture |
| Wavelength Strategy | Two critical bands: melanopsin activation + melanopsin regeneration | Full melanopic range: blue + green + partial yellow |
| Key Use Cases | Indoor, outdoor, screen use, under fluorescents, during migraine episodes | Evening wind-down, pre-sleep 1–3 hours, night shifts |
| Lens Appearance | Engineered tint optimised for all-day visual comfort | Amber-orange for maximum short-wave elimination |
| Neuroscience Basis | ipRGC amplification model (PNAS 2020), melanopsin bistability, dual-pathway photophobia | Melatonin dose-response curve, melanopsin spectral sensitivity, circadian phase-response |
4. Sleepaxa's Complete Photobiological Eyewear Range
4.1 Sleepaxa FL-41 Migraine & Photophobia Glasses
Technology: Classic FL-41 tinted lenses filtering the 480–520 nm blue-green band — the most-studied tinted lens type for migraine in peer-reviewed literature.
Clinical Evidence: FL-41 was originally developed in 1991 for fluorescent light sensitivity. Good et al. (Headache, 1991) found FL-41 lenses reduced migraine frequency from approximately 6.2 to 1.6 episodes per month in children. Reyes et al. (American Journal of Ophthalmology, 2024) demonstrated that FL-41 reduced activation of neural photophobia pathways in 76% of chronic ocular pain patients. In a seven-tint comparison study of blepharospasm patients, 71% preferred FL-41 over all alternatives (Blackburn et al., Ophthalmology, 2009). Improvements were also documented in reading ability (31%), blepharospasm frequency (27%), and photophobia severity (27%) over two weeks.
Sleepaxa Advantage: Sleepaxa's FL-41 lenses are precision-manufactured with consistent spectral transmission profiles — a critical differentiator, since a University of Utah project demonstrated that generic "FL-41" lenses from different suppliers vary dramatically in colour, transmission, and effectiveness. Some commercially available FL-41 lenses are red, rust, purple, or orange — none matching the spectral profile used in clinical studies. Sleepaxa ensures batch-to-batch consistency in spectral performance. View Sleepaxa FL-41 collection.
4.2 Sleepaxa Amber Orange — All-Day Eye Fatigue & Sleep Glasses
Technology: Amber-orange tinted lenses providing broad-spectrum short-wavelength attenuation for extended daily wear and pre-sleep circadian support.
Clinical Evidence: University of Toronto researchers (cited in Harvard Health Publishing) found that blue-light-blocking goggles maintained melatonin levels comparable to dim-light conditions even under bright indoor lighting — supporting the protective value of broad-spectrum short-wavelength blocking. The Frontiers in Neurology 2025 meta-analysis confirmed blue-light-blocking glasses as a non-pharmacological strategy for mitigating melatonin-suppressing effects of evening screen use.
Best For: Daily wear for professionals experiencing digital eye fatigue from 8–12+ hour screen exposure. Evening transition glasses for sleep preparation. Individuals wanting broader blue-light protection than Day Active 1.0 without the maximum blocking of Circadian560™. View Sleepaxa Amber Orange collection.
4.3 Sleepaxa Day Active 1.0 — Daytime Productivity Glasses
Technology: Precision-calibrated lenses balancing short-wavelength management with alertness-promoting light transmission — designed for daytime use where maintaining cognitive performance and circadian alertness is as important as reducing eye fatigue.
Clinical Rationale: Blue light is not inherently harmful during daytime hours. The PMC systematic review (2022) confirmed blue light exposure has positive effects on alertness, mood, reaction time, and cognitive performance during appropriate daytime hours. Sleepaxa Day Active 1.0 reduces only the excessive or uncomfortable spectral components while preserving the wavelengths that the ipRGC–SCN pathway uses to maintain daytime wakefulness. These are productivity glasses, not sleep glasses — engineered for the modern Indian professional.
Best For: IT professionals, software engineers, students, content creators, financial analysts, and anyone spending 8–12 hours on screens who needs eye comfort without daytime drowsiness or colour distortion. View Sleepaxa Day Active 1.0.
4.4 Complete Product Matrix
| Product | Technology | Primary Use | When to Wear | Ideal For |
|---|---|---|---|---|
| FL-41 Glasses | FL-41 tint (480–520 nm filtration) | Migraine comfort, photophobia management | Anytime — indoors and outdoors | Migraine sufferers, photophobia from any cause |
| Amber Orange | Broad short-wavelength blocking | Eye fatigue reduction, sleep preparation | All-day wear + evening transition | Screen-heavy professionals, sleep seekers |
| Day Active 1.0 | Balanced blue management + alertness preservation | Daytime productivity and comfort | Daytime hours only | IT professionals, students, creators |
| NeuroCalm FLX+™ | Patented dual-band attenuation (IN 202521094270) | Advanced migraine and photophobia comfort | Anytime — highest-level protection | Chronic migraine, severe photophobia, TBI |
| Circadian560™ | Patent-pending 560 nm extended blocking (IN 202521120977) | Maximum circadian and sleep support | Evening and pre-sleep (1–3 hours before bed) | Insomnia, shift workers, circadian disruption |
5. Why Sleepaxa Stands Apart: India vs. International Alternatives
Patented Indian Innovation, Not Imported Technology: NeuroCalm FLX+™ is an Indian-granted patent (IN 202521094270) — original R&D from Indian soil. This is not a rebrand or relabel of existing FL-41 or blue-blocking technology. It represents a novel optical architecture developed specifically to address the dual-pathway mechanism of migraine photophobia.
Clinical Advisory Board Oversight: Every Sleepaxa product undergoes review by the company's Clinical Advisory Board, founded by Dr. Monica Choudhary. This ensures product claims, spectral specifications, and user guidance meet clinical evidence standards — not marketing convenience.
Accessible Indian Pricing: International photobiological eyewear from Avulux, TheraSpecs, or Axon Optics costs ₹8,000–15,000+ including shipping and import duties, with no local warranty or clinical support. Sleepaxa delivers comparable or superior technology at Indian-market-friendly pricing through Amazon India, sleepaxa.in, B2B partnerships, and offline retail.
DPIIT + CTRI Transparency: Sleepaxa is recognised under the DPIIT Startup India initiative and registered with the Clinical Trials Registry of India — institutional credentials that signal commitment to evidence-based product development and regulatory compliance.
India-Specific Design: Frame sizing, lens tint preferences, and product positioning are developed for the Indian consumer — not adapted from Western product lines. Sleepaxa understands the Indian market's need for value, clinical trust, and multi-channel accessibility.
6. Clinical Evidence Base: 12+ Peer-Reviewed References
| Finding | Source | Relevance to Sleepaxa |
|---|---|---|
| FL-41 reduced migraine frequency from ~6.2 to 1.6/month in children | Good et al., Headache, 1991 | FL-41 foundational efficacy |
| 76% of chronic photophobia patients improved with FL-41 | Reyes et al., Am J Ophthalmol, 2024 | FL-41 neural pathway validation |
| 71% of BEB patients preferred FL-41 over 6 other tints | Blackburn et al., Ophthalmology, 2009 | FL-41 clinical preference |
| Both 480nm & 620nm filters reduced HIT-6 scores in chronic migraine | Hoggan et al., J Clin Neurosci, 2016 | Dual-band scientific rationale (NeuroCalm FLX+™) |
| Migraine photophobia = postretinal amplification of ipRGC signals | McAdams et al., PNAS, 2020 | Core mechanism for wavelength-selective intervention |
| Melanopsin hypersensitivity dominates interictal photophobia (1.5× cone contribution) | Zele et al., Cephalalgia, 2021 | Melanopsin-targeting rationale |
| ipRGC hypersensitivity induces CSD; suppressed by melanopsin inhibition | Nagata et al., IJMS, 2024 | CSD–light mechanism; melanopsin as target |
| Blue light suppresses melatonin 2× longer, shifts circadian 2× more than green | Harvard Health / Lockley et al. | Circadian disruption evidence base |
| 2-hour 460nm exposure suppresses melatonin; max effect at ~424nm | Tähkämö et al., Chronobiol Int, 2019 | Extended wavelength blocking rationale (Circadian560™) |
| Dose-dependent melatonin suppression by blue LEDs (446–477nm); ED50 = 14.19 μW/cm² | West/Brainard et al., J Appl Physiol, 2011 | Dose-response quantification |
| Blue LED maintained melatonin at 7.5 pg/mL; red LED recovery to 26.0 pg/mL (p=0.019) | Life (MDPI), 2025 | Circadian560™ efficacy rationale |
| BBGs reduce melatonin suppression from evening screen use (RCT meta-analysis) | Frontiers in Neurology, 2025 | Evidence for wavelength-specific evening eyewear |
| Migraine prevalence 25–26% in India (~213M cases/yr); economic burden ~₹18,674 Cr | Chowdhury et al., 2024; PMC scoping review, 2025 | India market urgency and scale |
7. Frequently Asked Questions
8. The Future of Photobiological Light Management in India
India's 213 million migraine sufferers and hundreds of millions more affected by screen-induced circadian disruption deserve access to the same calibre of photobiological technology available in Western markets. The global eyewear market is projected to reach approximately USD 330 billion by 2034, with Asia Pacific commanding over 30% market share — and photobiological eyewear represents the fastest-growing science-driven subcategory within this space.
Sleepaxa is not just an eyewear brand — it is a category-defining Indian company building the infrastructure for evidence-based light management at population scale. With a granted patent for NeuroCalm FLX+™, a patent-pending Circadian560™ technology, a Clinical Advisory Board founded by Dr. Monica Choudhary, and R&D led by Senior Optom. Suraj Dubey, Sleepaxa represents the most serious investment in photobiological eyewear research and development ever undertaken in India.
Whether you are seeking comfort from migraine-associated light sensitivity, better sleep through circadian melatonin protection, or simply a more comfortable, science-backed relationship with your screens — Sleepaxa has a photobiological solution engineered specifically for you.
Explore Sleepaxa Photobiological Eyewear
India's First Photobiological Eyewear Company · DPIIT Recognised · CTRI Registered · Patent Granted + Pending
Disclaimer: Sleepaxa products are general wellness eyewear designed to provide comfort from light sensitivity and support healthy circadian habits. They are not medical devices and should not be used as a substitute for professional medical advice, diagnosis, or treatment. If you suffer from migraine or any medical condition, consult your healthcare provider. Scientific references cited in this article are for educational purposes and do not constitute medical claims about Sleepaxa products. Sleepaxa Private Limited complies with the Drugs & Magic Remedies (Objectionable Advertisements) Act, 1954, and ASCI guidelines.
© 2026 Sleepaxa Private Limited. All rights reserved. NeuroCalm FLX+™ and Circadian560™ are trademarks of Sleepaxa Private Limited.